What is 8% and what is 10%? Are they the same type of water softener resin? Do you keen on these points? Then let us begin.
Resins that use for water treatment can naturally originate and synthetically originate. Natural resins can find in various geological environments, such as deposits of volcanic ashes in marine and lakes and sedimentary rocks. Zeolite is one of the resins that appears in nature. Clay, Dolomites are other examples of naturally occurring resins. Humankind prepared these resins artificially. Today, the world uses synthetic resins for many purposes, and water treatment is one of the largest fields that utilize resins.
Do you know the origin of synthetic water softener resins?
Water softener resins were developed with two monomers, and the resulted beads are strong and have great exchange capacities. Although the resin manufacturing components are still not changed in the resin industry but are modified to meet goals in several applications.
Ion exchange, a reversible chemical reaction where dissolved anions or cations are replaced with its relevant cation and anion, respectively, is the method used in resins to remove hardness in water.
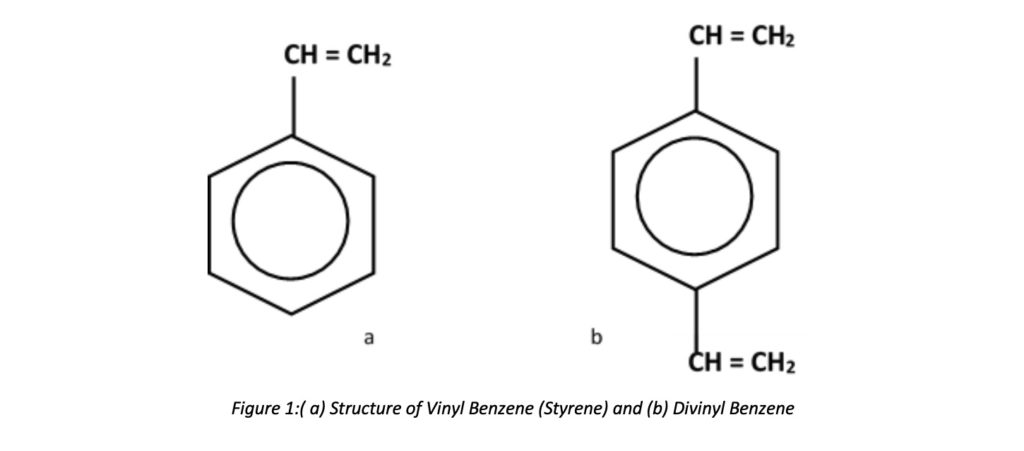
The most common materials of construction of Resins (softener resins) are Styrene (Vinyl Benzene or S) and Divinyl Benzene (DVB). The DVB is the binding agent, and it holds the resin together. It is also styrene and has a reactive vinyl (-HC=CH2) on both ends. DVB amount used while manufacturing can define as cross-linking percentages. It simply describes that the resin bead strength depends on DVB. Styrene and DVB are monomers in liquid form. But they are not dissolvable in water. While agitating, the monomers and water create small tiny monomer droplets suspended in the water phase. These small liquid droplets become hard plastic spheres (beads or copolymers) with proper agitation of the mixture and raising temperature. Both Cation and anion resins follow a similar process, but various components can use. The beads are washed, dried, and screened. After that, beads are functionalized (electrostatically charged) into an ion exchange resin. Kinetic performance of resins, resistance to oxidation, and service life of beads are mainly affected by the level of DVB. For example, approximately 2- 20% DVB content can be varied while manufacturing strong acid cation (SAC) exchange.
What is expressed as a percentage?
Styrene and DVB are combined, the percentage of DVB used can be expressed through these percentages. S/DVB combination takes different mixing ratios. There are different cross-linked percentages as 6%, 8%, 10%, and 15%.
This means that a high amount of DVB carries a high cross-link percentage. Usually, DVB is more expensive than styrene. Therefore, with an increasing rate, the cost of the resin is getting increased. DVB supplies mechanical stability to the resin beads.
The level of cross-linking directly causes moisture and plastic content within the beads.
8% cross-linked resins are the standard within the industry. But due to the application, in most cases, 10% of cross-linked resins are also in use.
Which one is better, 8% or 10%?
Let’s find the answer by introducing points.
- DVB content in 8% cross-linked resin is lesser than 10% cross-linked resins. The contained DVB amount of resin beads directly affects the moisture content, plastic content, and resin capacity.
- Those oxidants cause attacks on polymer cross-links, exposing them to a high oxidizing environment and shortening the lifespan. With increasing cross-linked percentage, resins capacity is also growing. However, the moisture content decreases—high moisture content results in less tolerance to chlorine. So 10% of cross-linked resin has high performances with a chlorine environment. Few chances are available to break down the beads with chlorine.
- Resins have more durability and strength with high percentages.
- Resins with high percentages are harder than low percentage resins. So 10% of cross-linked resins are more rigid than 8% resins.
- Also, with the high amount of cross-links, the resins are resisted with iron and manganese. Hence it may help to reduce resin fouling.
- With the above points, a high amount of DVB in resin manufacturing results in an extended lifetime of the resins.
- Softener resins need to be regenerated to make them more efficient. 10% resins are more comfortable with high frequencies of regeneration.
The data clearly shows that 10% of cross-linked resins are performed well than 8% of resins. But, if you have well water only with hardness, no iron, no manganese, and no high chlorine content, then it is perfect for selecting 8% resins. Though there is increased resistance to chlorine in 10%, it permits the resin oxidation and breakdown of the resins with time passes.
Both 8% and 10% resins are the bests for your softener. But, please consider your water quality before selecting the resins.
Whatever the cation resins with different cross-link percentages are getting degraded due to the de-crosslinking, and lead to increase the swelling and water retention capacity with the time.
Are there any drawbacks with high and low levels of cross-linking?
Crosslinking gives advantages, but also there are some disadvantages too. High cross-links have less ability to water holding; therefore, it congests the pathways in the resin beads. Also, regeneration is much more difficult. Resins exhibit a low operating capacity as a result of the high cross-linking. It may require high brine concentration for renewal and can decrease selectivity. But lower levels produce more open spaces in resin beads. It has adequate selectivity for hard water cation exchange. Not like higher levels, low levels can regenerate easily. The downside of the low cross-link resins is fewer capacities and less strength. So they are unable to handle the excess flow rates physically. Resin breakages, losses, and pressure losses can develop in deformation occurs with water with high temperatures. In addition, they are very much sensitive to oxidizing agents.
Conclusions
- Water softener resins can find in nature. But artificial resins are most popular in the water treatment industry. We can control the physical and chemical properties of resins during resin manufacturing.
- S/DVB combination is the common monomer combination that uses for softener resin manufacturing. But it can differ with the combined ratio to achieve the specific targets.
- After a long process, these polymers are converted to electrostatically charged resin beads.
- There are cationic resins and anionic resin. In the water softening industry, it is are commonly used cation resins.
- While manufacturing, structural changes of resins can be identified as cross-links, which occur due to DVB dosage. DVB is more expensive than styrene; therefore, resins with high cross-linked percentages (which contain more DVB than low levels) are more expensive.
- With increasing levels of cross-links, there are benefits and a few drawbacks.
- 8% is the standard level of resins. These 8% cross-linked resins have been used over the years for household water softening applications. It shows the best combination in softening, representing the best durability and kinetics.
- 8% or 10% (High or Low cross-linked percentages) should be considered in special cases, such as high-temperature levels, high oxidant levels (chlorine/chloramines), and in the presence of iron and manganese levels in the water.
- You can select either 8% or 10% as your softener resins, but be open-minded to consider all the points for better performance of softening plant. Select the best product, not the high-cost product.



One Response
Excellent explanation of the 8% & 10% of the cross linkes.
Thank you.