DI represents “Deionization.” DI process removes all impurities or negative and positive ions that dissolve in water. Deionization produces high quality purified water for many applications.
Running a DI plant is much more expensive, but it is cost-effective with ion exchange resins. Ion exchange resins support the primary function in the water deionization (DI) process. This article will help you get full basic knowledge of the functioning of ion exchange resins.
Are ion exchange resins and mixed bed resins (MB) identical?
A beginner in the resin industry probably messed up with ion exchange resins and mixed bed resin. But it is simple, and follow the article to learn more.
What are resins?
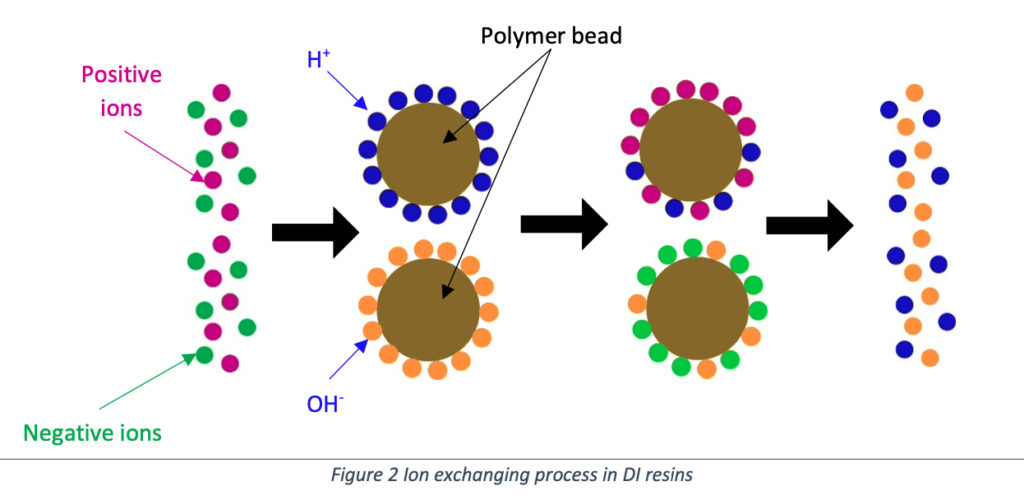
Resins are manufactured with styrene crosslinking with divinyl benzene (DVB). They are tiny, spherical shaped plastic beads. Permanently attached fixed negative and positive charge functional groups are contained in plastic beads’ polymer structures. When the functional group is positive, the bonded ion is negative, and then we call that anion resin. At the same time, the functional group is negative, and the bonded ion is positive. We call it cation resin.
There are four types of resins; Strong Acid Cations (SAC), Weak Acid Cations (WAC), Strong Base Anions (SBA), and Weak Base Anions (WBA). The mainly used resin types for the deionization process are Strong Acid Cations and Strong Base Anions.
SAC resins contain a sulfonate group as its functional group and are present in the form of Hydrogen ions (H+). The quaternary ammonium group is the functional group of SBA resins and is present in the form of Hydroxyl ions (OH–).
What are mixed bed resins?
Mixed bed resins are a combination of strong acid cations and strong base anions in a 2:3 ratio.
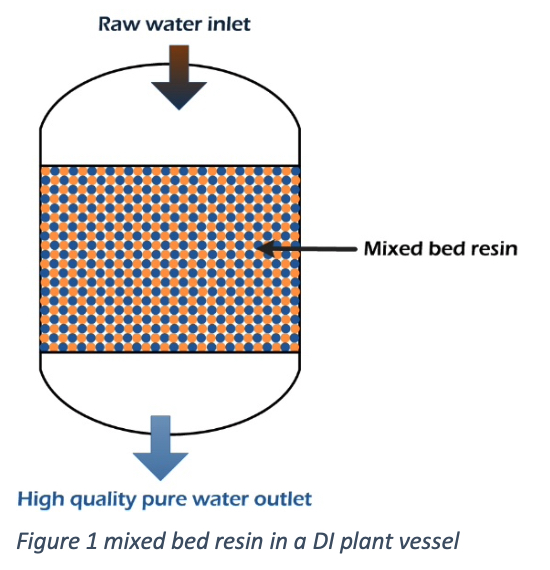
Conventional deionization plants use two beds; one vessel contains a cations resin bed, and the other has an anion resin bed separately.
The resins always act as the ion exchange media in water treatment processes. So the two different ion exchange resins are mixed in one tank and prepared in one bed; it is called mixed bed resin. The MB resins also exchange ions. Ion exchange resins are manufactured in various forms. It will be the difference in resins used in multiple applications; softener resins appear in the form of Na+ and the deionizing cation resins present in the form of H+.
The mixed bed resin working process
A mixed bed deionizer has both anion and cation resins in one single unit. The ion exchange process will be repeated in thousand times with the water flow through the MB resin bed. It helps to produce high quality pure water by the DI plant.
The raw water with dissolves positive and negative ions enters the vessel, and flows through the mixed bed. Water can consist of Ca2+, Mg2+, Na+, HCO3-, SO42-, Cl–, and NO3– ions. The mixed bed contains anion and cation resins.
Cation resins with the form H+ have replaced their H+ ions with all positive ions in raw water and released H+ ions.
R – H + Ca2+ → R – Ca + H+
Anion resins with the form of OH-, exchange its hydroxyl ions with the negative ions in water and release hydroxyl ions.
R – OH + NO3– → R – NO3 + OH–
Same manner, both anion, and cation resins release OH– and H+ ions, and deionized water will be generated. Following examples shows you the theory in a simple manner.
Mixed bed resins eventually get depleted, and we can re-activate the resin bed to work properly. The reactivation process is called “regeneration .”The complicated process of the mixed bed resin plant is regeneration.
How to regenerate a mixed bed resin plant?
Regeneration takes a few steps to complete its procedure.
Backwashing
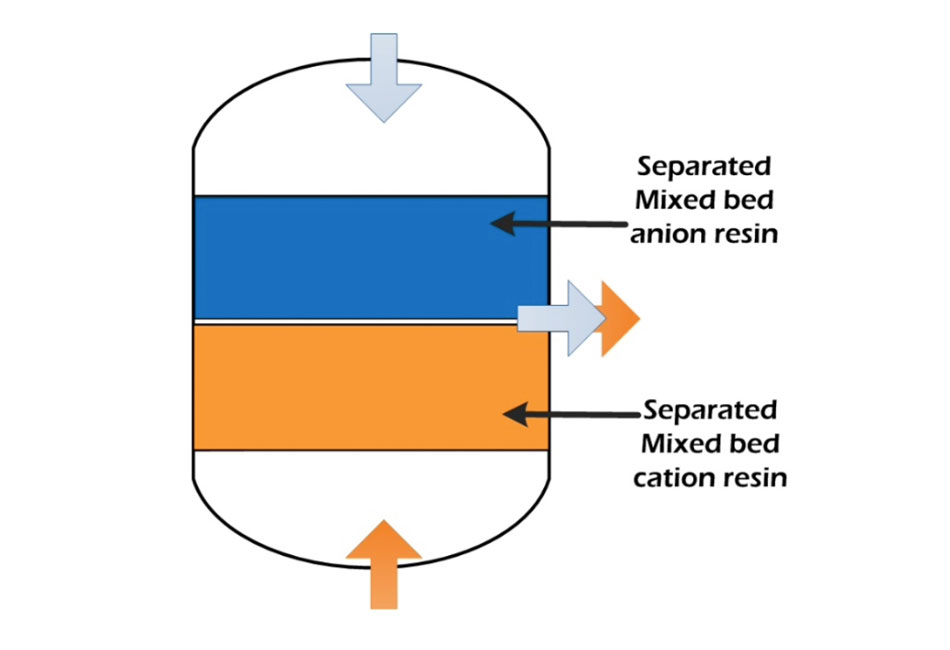
Backwashing is the first step, and backwashing the exhausted MB results in two separate anion and cation phases according to their density. The cation resin has a higher density than anion resin; therefore, cation resin goes down, and anion resin goes upstream. Backwashing flow rates need to be adjusted to have enough bed expansion. Also, anion resins can be entrapped at the bottom layer; therefore, changing flow rates helps prevent accumulation at the cation resin layer. It is necessary to start with a high flow rate for fluidization of the resin bed, and reducing the flow rate will prevent the anions from escaping. Backwashing can be conducted within 20-30 minutes, but it can be 40 – 50 minutes with the heavy suspended solid load.
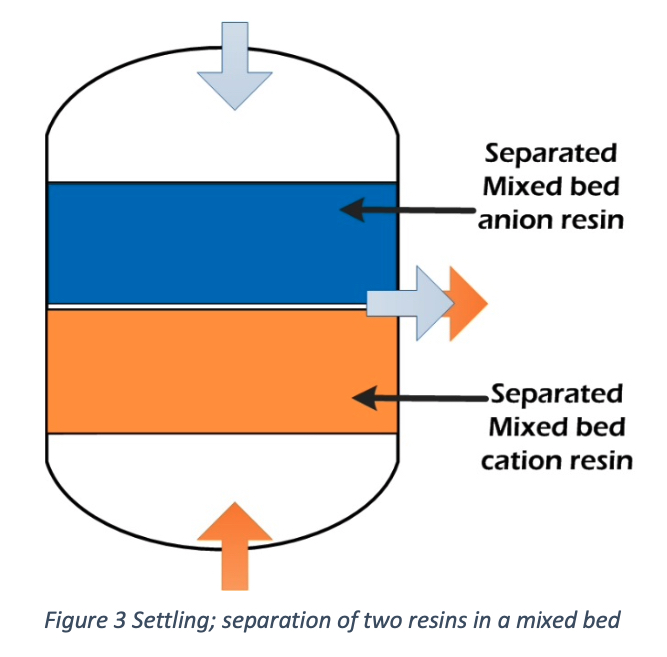
Settling
After Backwashing, it will take a few minutes to settle the anion and cation resins into separate layers. We can observe a sharp separation line, and no more cross mixing of anions and cations is visible.
Dosage of regenerant
Caustic soda (NaOH) is a commonly used regenerant for anion resins, and hydrogen chloride (HCl) is the regenerant for the cation resins. NaOH is injected from the top of the vessel and collected from the middle collector. HCl is injected at the bottom of the line and collected from the middle collector. Usually, the injection time should be 20-30 minutes. It would be best if you used prescribed concentrations for acid and base.
Regenerant injection can be arranged in “simultaneous injection” and “sequential injection.”
- Simultaneous injection: Acid and the base are injected at the same time.
- Sequential injection: If you are not possible to inject acid and the base simultaneously, you can start with caustic soda (the base). While using the base, there should be a buffer flow of water at the bottom to prevent the caustic contamination of the cation resin layer. After completing the base injection and the displacement, the acid must be injected into the cation resin bed. There should be a small water flow rate at the anion resin bed to prevent acid contamination of the anion resin bed.
Slow rinsing
When you complete the regenerant introduction to the resin beds, continuous water flow is required to displace the regenerants.
Draining
The water in the shell needs to be drained before occurring air mixing. It will help to have a homogenous mixing of both anion and cation resins. The water is requested to drain down up to the level of the resin bed. Please follow the suppliers’ guidelines.
Mixing
The recommended airflow help anion and cation resins beds mix with nitrogen or fresh air. There is a chance to trace oil when you use a compressor. Mixing time can be >5 minutes and less than 15 minutes.
Water filling
At the top of the vessel, a water flow can be inserted into the tank. It will take some time to fill with water. You can use NaOH diluted water in the first few minutes with the support of a feedwater line at a service flow rate to prevent the disturbance of the mixing of resins.
Final rinsing
This step also can identify as the fast rinsing, and it is used to complete the regeneration procedure to obtain the required effluent quality of the DI plant.
Conclusion
Discussing mixed bed resin activities is pretty interesting with the basic knowledge. What is Deionizing? What is the process of DI? What are DI resins? How do resins get prepared? Do I have an idea of mixed bed resins?, and How does mixed bed resin work? Find your answers by reading this article. Here is a brief of the above.
- Removing all impurities in water and producing high quality water can be introduced as Deionization.
- Deionized water can be generated with different methods, but we discussed the ion exchange method here.
- Anion and cation resins electrostatically bonded with hydroxyl and hydrogen ions, respectively, are used in the ion exchange process.
- Mixed bed resins combine the above anions and cations in a selected ratio.
- Mixed bed resins are more effective than two bed DI plants.
- Mixed bed resins can eventually get depleted and need to regenerate.
- Regeneration can be introduced in “simultaneous injection” and “sequential injection.”
- Regeneration has some steps to complete the entire process, and they are, Backwashing, settling, regenerant introduction, Slow rinsing, Mixing, Water filling, and fast rinsing.